
Bludigo® Demonstrated Fast and Reliable Visualization of Ureter Flow
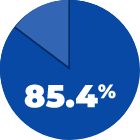
85.4% of ureter patency assessments reported color contrasts in jet stream ranging from significant  to striking
to striking  1
1
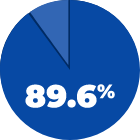
89.6% of patients were defined as a responder2,a
FAST DETECTION
Within 4-9 minutes
post-IV injection1
Urine jet conspicuity score by the blinded central review process was assessed using the following 5-point ordinal scale: 1 = No jet observed; 2 = Weak jet, little color contrast; 3 = Color contrast or significant jet flow; 4 = Strong jet flow with good color contrast; 5 = Strong jet flow with striking contrast in color.1
*An exploratory analysis showed no statistically significant difference between the 2 doses of indigo carmine in providing visualization of urine efflux.1
aA patient was considered a responder when the Bludigo® conspicuity score was ≥3 and a ≥1 increase compared with placebo.2
|
||
95% CL | (N=49) | (N=49) |
95% CL‡ | (48%, 77%) | (61%, 87%) |
95% CL | (63%, 88%) | (68%, 91%) |
95% CL | (57%, 83%) | (68%, 91%) |
†A subject was a responder when there was ≥1-point improvement in the conspicuity scores following the indigo carmine vs saline treatment (indigo carmine – saline ≥1) and the conspicuity score following the indigo carmine treatment was 3, 4, or 5. The responder criteria were assessed separately for each ureter for each subject based on the blinded central reviewer’s conspicuity score.1
‡Two-sided 95% confidence limits for the proportion of responders, calculated using the Clopper-Pearson (exact) method.1
Study design: Bludigo® was studied in a phase 3, open-label, randomized, multicenter, parallel-group study of 118 patients. The study was composed of 3 periods: Screening (up to 30 days prior to surgery), Randomization/Dosing (Day 1), and a 30-day Safety Follow-up Period. Patients served as their own control by receiving a dose of normal saline prior to randomization, at which point patients were randomized 1:1 to receive 2.5 mL or 5 mL indigo carmine. The primary endpoint was efficacy based on a visualization conspicuity score. Efficacy was assessed using a 5-point ordinal scale measuring contrast flow. Secondary endpoints were physician satisfaction, time to visualization, and proportion of responders. No significant statistical difference was found between doses, and the 2.5 mL dose of Bludigo® is not approved or available in the United States.1
CL=confidence level; IV=intravenous.
BLUDIGO® (indigotindisulfonate sodium injection, USP)
INDICATIONS AND USAGE
BLUDIGO® is a diagnostic dye indicated for use as a visualization aid in the cystoscopic assessment of the integrity of the ureters in adults following urological and gynecological open, robotic, or endoscopic surgical procedures.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
BLUDIGO® is contraindicated in patients with known hypersensitivity to indigotindisulfonate or any of its components.
WARNINGS AND PRECAUTIONS
Cardiovascular Reactions: Severe or life-threatening cardiovascular reactions including cardiac arrest, arrhythmia, asystole, atrioventricular block second degree, hypotension, elevation in blood pressure, bradycardia, and tachycardia have been reported. Closely monitor blood pressure and cardiac rhythm during and following the BLUDIGO® injection. Interrupt administration if reactions are observed.
Hypersensitivity Reactions: Serious anaphylactic reactions with hypotension, dyspnea, bronchospasm, urticaria, or erythema have been reported. Monitor patients for anaphylactic reactions and have emergency equipment and trained personnel readily available.
Interference with Oximetry Measurements: Anesthesiologists should be aware of the potential for artifactual reduction in SpO2 when anesthetized patients are administered BLUDIGO®.
USE IN SPECIFIC POPULATIONS
Renal Impairment: BLUDIGO® is not recommended for use in patients with eGFR<30 mL/min.
Pediatric Use: The safety and effectiveness of BLUDIGO® have not been established in pediatric patients.
Pregnancy and Lactation: Please consult the Full Prescribing Information before using BLUDIGO® in a patient that is lactating, pregnant, or may be pregnant.
RECOMMENDED DOSAGE
The recommended dose for BLUDIGO® is 5 mL given intravenously over 1 minute.
IMPORTANT ADMINISTRATION INSTRUCTIONS
- Monitor blood pressure and cardiac rhythm during and following the injection.
- Use immediately after opening ampule.
- Withdraw the contents of the ampule through a 5 micron or smaller filter straw/filter needle to ensure that the withdrawn solution contains no particulates. The withdrawn solution should be inspected visually for particulate matter and discoloration prior to administration.
- Do not administer with infusion assemblies used with other diluents or drugs.
- Discard any unused portion.
ADVERSE REACTIONS
Clinical Trial Experience: The most common adverse reactions (1%) associated with BLUDIGO® in clinical trials were: constipation, nausea, vomiting, abdominal pain, pyrexia, ALT increase, and dysuria.
Postmarketing Experience: The following adverse reactions have been identified following the use of indigotindisulfonate sodium injection products:
- Cardiovascular disorders: cardiac arrest, arrhythmia, asystole, atrioventricular block second degree, hypotension, elevation in blood pressure, bradycardia, tachycardia
- General disorders and administration site conditions: injection site discoloration
- Immune system disorders: anaphylactic reactions with hypotension, dyspnea, bronchospasm, urticaria, erythema
Please see the full Prescribing Information for additional Important Safety Information.
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
References: 1. Data on file. PVP-10ICO1 clinical study report. Newark, NJ: Provepharm, Inc.; 2025. 2. Boytim M, Jones MJ, Bart AS, Svintozelskiy P, Albala DM. Comparison of methods for ureteral patency visualization. Rev Urol. 2024;23(1):e67-e74.




